Toothpaste Regulation: New Rules and Compliance Guide
or complex compliance issues.
clearance and fund security.
With the continuous development of the toothpaste market and the diversification of consumer demand,the formulation and implementation of relevant regulations and standards have become increasingly important.Recently,The registration and filing information service platform has been updated,adding a "Toothpaste User" section,which is undoubtedly a signal of upcoming new regulations in the toothpaste industry.This article will delve into the details of these new regulations,their requirements,and how companies should respond.
I.Updates to the Registration and Filing Platform
1.Added the "Toothpaste Users" section Output:
The Cosmetics Registration and Filing Information Service Platform has been updated,with a new "Toothpaste User" section added under the "Enterprise Information Management" category.This change signals the imminent implementation of new regulations for toothpaste,which undoubtedly adds an additional layer of compliance requirements for toothpaste manufacturers and sellers.
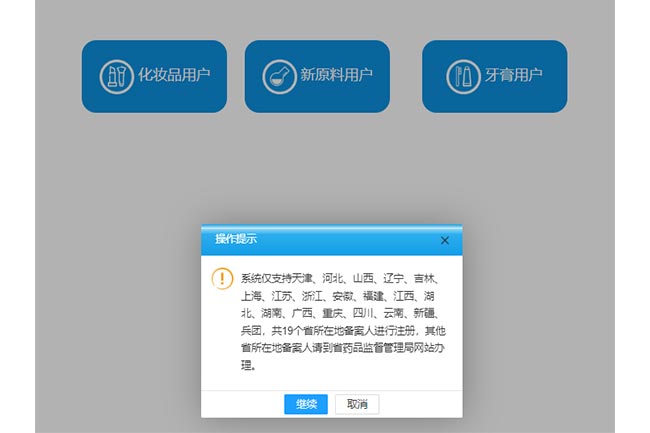
Restrictions on Application Provinces Output:
The newly added "Toothpaste User" section restricts the filing to specific provinces.Registrants from 19 provinces,including Tianjin,Hebei,Shanxi,Liaoning,Jilin,Shanghai,Jiangsu,Zhejiang,Anhui,Fujian,Jiangxi,Hubei,Hunan,Guangxi,Chongqing,Sichuan,Yunnan,Xinjiang,and the Xinjiang Production and Construction Corps,can register on this platform.Registrants from other provinces must complete the process on their respective provincial Drug Administration websites.
Preparation of Filer Materials Output:
Both domestic and overseas filers must prepare a series of documents for registration,including a filer information form,quality and safety responsible person information,quality management system overview,adverse reaction monitoring system overview,etc.Overseas filers additionally need to provide an authorization letter,manufacturer information form,GMP documents,etc.
II.Published Toothpaste Regulatory Requirements
Toothpaste Supervision and Management Measures Output:
Currently,published toothpaste regulations include the Toothpaste Supervision and Management Measures and the Cosmetics Safety Technical Standards revision project,among others.These regulations provide clear guidance for the production,sale,and use of toothpaste.
Addition of Testing Items Output:
Regarding physicochemical testing items,toothpaste is similar to cosmetics but includes additional tests for hard particles,fluoride content,diethylene glycol,and ethylene glycol.These testing items are stipulated in the national toothpaste standards.
Standardization of Efficacy Claims Output:
For efficacy claims,the Toothpaste Filing Documentation Standards (draft for comments) limits the scope of claims toothpaste can make.Companies should use cautious language in efficacy claims to ensure compliance with regulatory requirements.
III.Toothpaste Regulations Worth Attention
Toothpaste Ingredient Catalog Output:
The Catalog of Used Toothpaste Ingredients will be used to determine whether new ingredients are used and standardize ingredient INCI names and Chinese names.This will help ensure the safety and quality of toothpaste products.
Toothpaste Labeling Management Measures Output:
The Toothpaste Labeling Management Measures will clarify mandatory labeling requirements for toothpaste.It is reported that the measures will be formulated with reference to the Cosmetics Labeling Management Measures,providing clear guidance for toothpaste product labeling.
Toothpaste Safety Assessment Technical Guidelines Output:
The Toothpaste Safety Assessment Technical Guidelines will be used to assess the safety of toothpaste products,providing a scientific basis for their safe use.
IV.Corporate Response Strategies
Prepare Documentation in Advance Output:
With the new regulations set to take effect on December 1,companies have just over two months to prepare.Therefore,companies should prepare the necessary documentation in advance to ensure smooth registration and filing of toothpaste products once the new regulations are implemented.
Closely Monitor Regulatory Developments Output:
Companies must also closely monitor the release and updates of toothpaste-related regulations,staying informed of regulatory requirements to ensure their production and sales activities comply with laws and regulations.
Strengthen Product Quality and Safety Management Output:
Companies should strengthen product quality and safety management to ensure compliance with regulatory requirements and enhance market competitiveness.
Was this helpful? Give us a like!
Contact our experts for compliance audits, precise quotes, and one-stop customs support.

Recent Comments (0) 0
Leave a Reply